Description
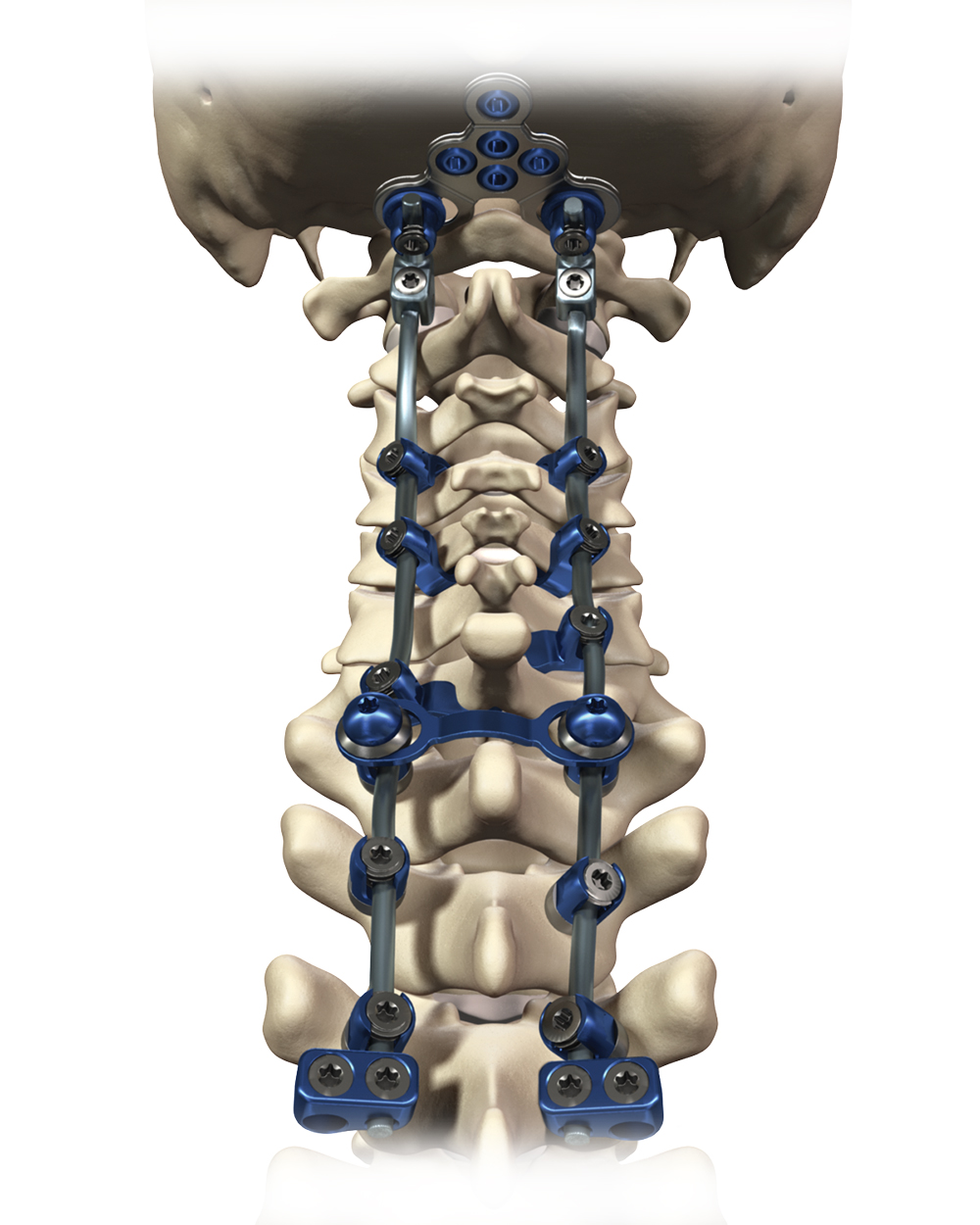
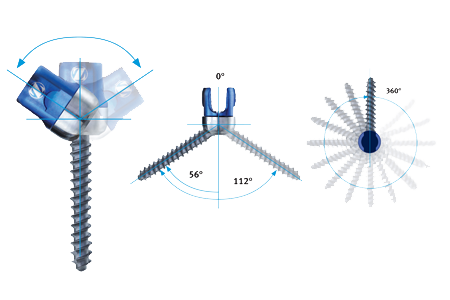
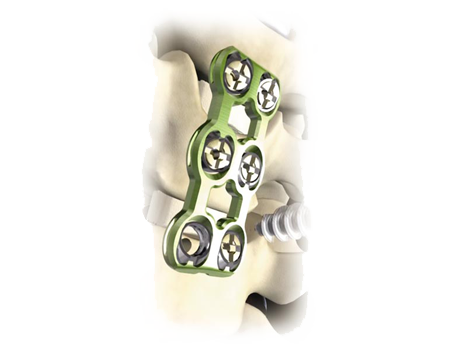
The Virage OCT Spinal Fixation System is a posterior system intended for the Occipital-Cervical-Thoracic spine (Occiput–T3). The system consists of a variety of rods, anchors, transverse connectors, screws, and polyaxial screws to achieve an implant construct as necessary for the individual case. The system also includes the instruments necessary for inserting and securing the implants. The implant system is intended to be removed after solid fusion has occurred.
The Virage System implants are fabricated from medical grade titanium alloy and medical grade cobalt chromium alloy. Implants made from medical grade titanium, medical grade titanium alloy, and medical grade cobalt chromium may be used together. Never use titanium, titanium alloy, and/or cobalt chromium with stainless steel in the same construct. All implants are single use only and should not be reused under any circumstances.
The titanium Lentur™ Cable System used with the Virage OCT Spinal Fixation System to allow for cable attachment to the posterior cervical or thoracic spine is optional and not approved in all countries.
Indications
The Virage OCT Spinal Fixation System is intended to provide immobilization and stabilization of spinal segments as an adjunct to fusion for the following acute and chronic instabilities of the craniocervical junction, the cervical spine (C1–C7) and the thoracic spine from T1–T3; traumatic spinal fractures and/or traumatic dislocations; instability of deformity; failed previous fusions (e.g., pseudoarthorsis); tumors involving the cervical spine; and degenerative disease, including intractable radiculopathy and/or myelopathy, neck and/or arm pain of discogenic origin as confirmed by radiographic studies, and degenerative disease of the facets with instability. The Virage OCT Spinal Fixation System is also intended to restore the integrity of the spinal column even in the absence of fusion for a limited time period in patients with advance stage tumors involving the cervical spine in whom life expectancy is of insufficient duration to permit achievement of fusion.
In order to achieve additional levels of fixation, The Virage OCT Spinal Fixation System may be connected to the Instinct Java and Sequoia Spinal Systems offered by Zimmer Spine, using rod connectors and transition rods. Refer to the Instinct Java and Sequoia Spinal System package insert for a list of the system specific indications of use.
The titanium Lentur™ Cable System to be used with the Virage OCT Spinal Fixation System allows for cable attachment to the posterior cervical or thoracic spine.
Contraindications
The Virage System is not designed or sold for any use except as indicated. DO NOT USE THE VIRAGE SYSTEM IMPLANTS IN THE PRESENCE OF ANY CONTRAINDICATION.
Contraindications include, but are not limited to:
- Overt infection or distant foci of infections.
- Local inflammation, with or without fever or leukocytosis.
- Pregnancy.
- Morbid obesity.
- Rapid joint disease, bone absorption, osteopenia, and/or osteoporosis.
- Suspected or documented metal allergy or intolerance.
- Any time implant utilization would interfere with anatomical structures or expedited physiological performance, such as impinging on vital structures.
- Severe comminuted fractures such that segments may not be maintained in satisfactory proximate reduction.
- Use in displaced, non-reduced fractures with bone loss.
- The presence of marked bone absorption or severe metabolic bone disease that could compromise the fixation achieved.
- Poor prognosis for good wound healing (e.g., decubitis ulcer, end-stage diabetes, severe protein deficiency, and/or malnutrition).
- Any case not needing a bone graft or fusion.
- Any case not described in the indications.
See the Warnings and Precautions section.